COM – Cell4Chem ou comment utiliser les bactéries pour valoriser nos déchets ?
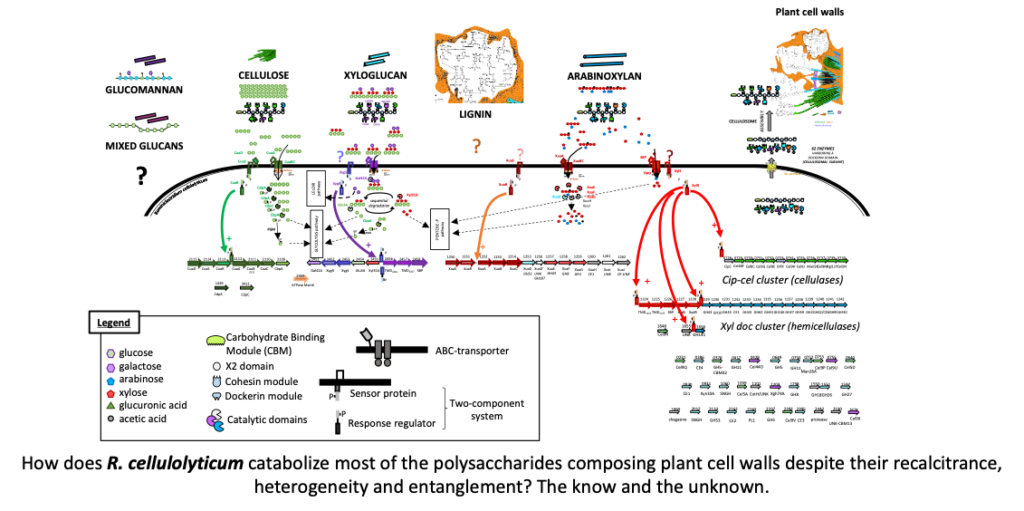
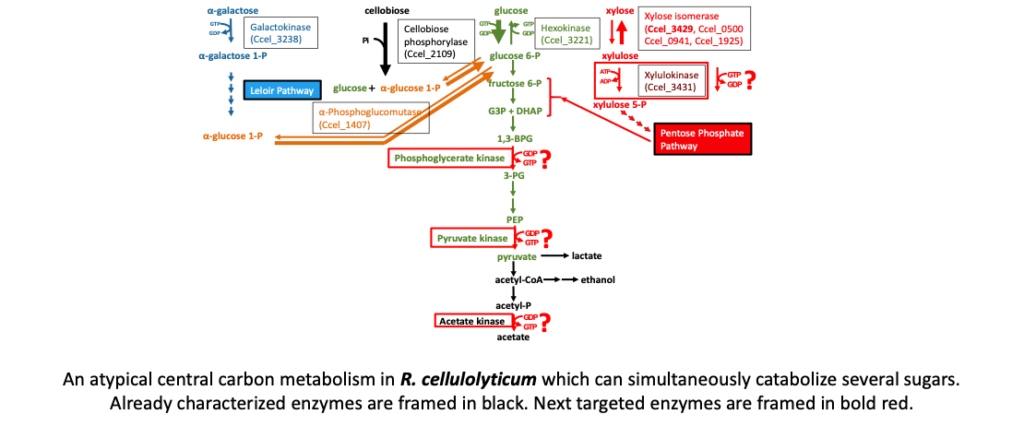
Cell4Chem est un projet Européen qui vise à produire des molécules d’intérêt industriel telles que les carboxylates à chaines moyennes, en utilisant un procédé écologique.